Office of Environmental Health and Safety
Bloodborne Pathogen Training for High Risk Personnel
Revised: September 4, 2018
Training Support
This training module must be taken during normal working hours so that you have ready access to Taylor Kriete, Biological Safety Officer. He is available at (504) 952-1337 to answer any questions you have related to this training.
Contents
- Introduction
- Regulations
- Exposure Control Plan
- Definitions
- Types of BBPs and Modes of Transmission
- Risk Determination
- Hepatitis B Vaccine
- Exposure Response
- Work Practices

I. Introduction
- The State of Louisiana Office of Risk Management requires that Bloodborne Pathogen (BBP) training be provided to personnel who can reasonably anticipate having contact with blood and other potentially infectious materials in the course of their duties.
- The U. S. Occupational Safety and Health Administration (OSHA) requires all personnel with occupational exposure to participate in a training program.
- This training includes hyperlinks(indicated by gold bold text) to provide you with additional information.
- Click here to view the OSHA standard.


You should read the LSUHSC Exposure Control Plan (ECP), which is designed to eliminate of minimize occupational exposure to BBPs. Its components include:
- Responsibilities.
- Exposure Determinations.
- Engineering and Work Practice Controls.
- Availability of the Hepatitis B Vaccination Series.
- Post-Exposure Actions.
School of Dentistry personnel should also be familiar with the additional guidance provided by the School of Dentistry ECP.
Definitions
- Blood includes human blood, human blood components, and products made from human blood.
- Bloodborne Pathogens are pathogenic microorganisms present in human blood and other body fluids that can cause disease in humans, including hepatitis B virus (HBV), hepatitis C virus (HCV), and human immunodeficiency virus (HIV).
- Occupational Exposure is reasonably anticipated skin, eye, mucous membrane, or parenteral contact with blood or OPIM that may result from the performance of the employee’s duties.
- Other Potentially Infectious Materials (OPIM) are materials other than human blood that can contain bloodborne pathogens and may potentially be infectious. OPIM include HIV-containing cell or tissue cultures or organ cultures and HIV- or HBV-containing culture medium or other solutions.
- Source Individual is any individual, living or dead, whose blood or other potentially infectious materials may be a source of occupational exposure.
- Standard Precautions is the use of personal protective equipment (PPE) to prevent exposure to both bloodborne and airborne pathogens.
- Universal Precautions is an approach to infection control in which all human blood and certain human body fluids are treated as if known to be infectious for HIV, HBV, and other bloodborne pathogens.
Examples of OPIM
Cerebrospinal, synovial, pleural, amniotic, pericardial, and peritoneal fluids; semen; vaginal secretions.
Any material fluid contaminated with blood; saliva in dental procedures.
Bodily fluids in emergency situations that cannot be recognized.
Unfixed human tissues or organs.
Unfixed tissues or organs from HIV- or HBV-infected animals.
HIV or HBV cell cultures or culture medium.
II. Types of BBPs and Modes of Transmission
HIV/AIDS
- AIDS is Acquired Immuno-Deficiency Syndrome, a fatal disease that diminishes the body's ability to fight infection; it is caused by the Human Immunodeficiency Virus (HIV).
- During the incubation period, which may last many years, there may be no signs an individual is infected with HIV; the person can, however, still pass the virus on to others.
- As the disease progresses, there may be flu-like illness, depressed immunity, and enhanced susceptibility to opportunistic infections.
- 682,668 infected in U.S.; an estimated 185 occupationally transmitted cases since 1981.
- Risk of HIV infection after a single positive needle stick is 0.3%. Currently no vaccine is available.
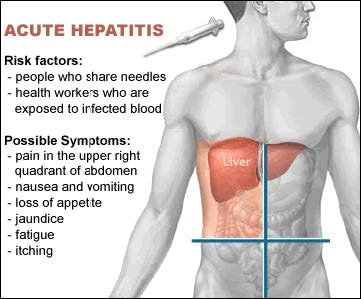
Hepatitis B Virus
- Hepatitis B is an infectious illness caused by the Hepatitis B Virus (HBV) which causes inflammation of the liver, liver infection, cirrhosis, and liver cancer.
- Hepatitis B is far more common than HIV and is present in very high concentrations in the blood of infected persons.
- Symptoms include flu-like illness, jaundice, dark urine, extreme fatigue, nausea, abdominal and joint pain, and rash.
- In 2007, overall incidence of reported acute HBV infection was 1.5 per 100,000 population.
- The risk of HBV infection after a single positive needle stick is 6% - 30%. A vaccination is available for Hepatitis B.
Hepatitis C Virus
- Hepatitis C is an infectious disease affecting the liver caused by the Hepatitis C Virus (HCV). Infection is often asymptomatic, but once established chronic infection can lead to liver fibrosis, cirrhosis, liver failure, and liver cancer.
- Early signs and symptoms include fatigue, fever, headaches, nausea, vomiting, loss of appetite, abdominal pain, jaundice, dark-colored urine, clay-colored stools.
- Estimated 3-4 million new HCV carriers, 175,000 documented cases, and 1,120 occupationally transmitted cases in U.S. annually. Risk of HCV infection after single positive needle stick is 3-10%. No HCV vaccine is available.
Modes of Transmission
- Transmission can be via direct and indirect contact.
- Transmission can occur through:
- Accidental punctures from sharp objects (i.e., needles, broken glass, or other contaminated sharps).
- Contact between broken or damaged skin and infected body fluids (e.g., open sores, cuts, abrasions, acne, or any sort of damaged or broken skin such as blisters).
- Exposure to mucous membranes, eyes, nose, or mouth.
- BBPs are not known to be transmitted across intact skin.
- Understand which routes are viable means of transmission for the BBPs in your work area and which are not.
III. Risk Determination
Exposure Risk Determination
Personnel are classified as high risk if they:
- Perform direct patient care activities likely to result in direct or indirect exposure to blood or body fluids.
- Process or handle human blood, body fluids, tissues or organs.
- Process or handle equipment, materials or waste that may be contaminated with human blood, body fluids or OPIMs.
- Routinely administer first aid.
- Have potential to be exposed to blood, body fluids or OPIM in their job duties.
- Examples: Physicians, dentists, laboratory workers, healthcare workers, animal care technicians, plumbers and custodial staff, shelter workers, child welfare workers, police officers and others who carry weapons, first responders, firefighters, kitchen staff (that may handle sharp equipment), and public safety workers.
Low risk personnel do not perform any activity listed above (e.g., clerical, administrative staff, IT).
Employee Risk Determination
- The Office of Compliance initially assigns risk levels and associated training for all employees based on job title and department.
- Each supervisor will validate the assigned risk level by reviewing tasks and procedures associated with the employee’s exposure to human blood, body fluids, or OPIMs.
- If the employee’s risk level changes due to supervisory review or the assignment of new tasks, notify the Biological Safety Officer so that the correct training module can be assigned.


Student Risk Determination
- Students enrolled in the Schools of Medicine, Nursing, Dentistry, and Allied Health Professions are classified as high risk and are required to be vaccinated for Hepatitis B.
- Students enrolled in the Schools of Public Health and Graduate Studies are initially classified as low risk.
- If, due to the nature of their work, a student in School of Public Health or Graduate Studies has potential exposure to human blood, body fluids or OPIM, that student will obtain the Hepatitis B vaccination form or complete the Hepatitis B Consent/Declination form(appendix A to the ECP) indicating that they decline the vaccination. Provide the immunization record or signed appendix Consent/Declination form to Student Health. That student will also have their classification changed to “high risk.”
IV. Hepatitis B Vaccine
- The hepatitis B vaccination series is available at no cost to all high risk employees.
- Vaccination is encouraged unless documentation exists that the employee has previously received the series, antibody testing reveals that the employee is immune, or medical evaluation shows that vaccination is contraindicated.
- The Centers for Disease Control and Prevention offers information on the hepatitis B vaccine’s efficacy, safety, method of administration, and the benefits of being vaccinated on their website.
- Employees may decline the hepatitis B vaccination, but must complete the Hepatitis B Consent/Declination form (Appendix A of Exposure Control Plan) indicating that they decline the vaccine. Note that employees who decline the vaccine may request the vaccine at a later date at no cost.
- The School of Nursing administers the Hepatitis B vaccine on a recurring basis.
- High risk employees should complete the Hepatitis B Consent/Declination form (Appendix A of Exposure Control Plan) and contact their supervisor/business manager, who will work with them to schedule an appointment.
- If you have any questions regarding the vaccine, contact Dr. Stephanie Pierce at Spierc@lsuhsc.edu or 568-4217.
V. Exposure Response
Exposure Incident
Exposure Incident: a specific eye, mouth, other mucous membrane, non-intact skin, or a puncture contact with blood or other potentially infectious materials that results from the performance of a person's duties.
For example:
- Needlesticks or scrapes and cuts with contaminated sharps.
- Contact with broken skin through cuts or rashes.
- Splashes to the eyes, nose or mouth.
If you’re not sure if you’ve had an exposure, check for punctures in your glove. If the glove is broken, assume an exposure has occurred.

Exposure Response
The following actions should be taken immediately after exposure:
- Administer first aid. Wash the needlestick or cut with soap and water. Notify your supervisor as soon as feasible.
- If exposure is by splashes of infectious materials to the nose, mouth or eyes, the affected area should be flushed extensively with water, saline or sterile irrigating solution.
- Document the routes of exposure, the biological material of exposure, and how the incident occurred.
- Seek medical attention as soon as possible. HIV prophylaxis is most effective if started within two hours of exposure.
After initial treatment is complete, make an appointment with your healthcare provider as soon as possible for appropriate follow-up.
For more information on exposure response actions, see Section 9 of the Exposure Control Plan .
Post-Treatment Actions
Following an exposure incident, the supervisor must report the incident and complete the appropriate reporting form(s) as outlined in the Incident and Accident Reporting and Investigation Policy.
Counseling for employees and students is available through the Campus Assistance Program (568-8888). The Student Health Clinic can provide access to the Expert Review Panel on behalf of students.
VI. Work Practices
Universal Precautions and Biological Safety
- Universal precautions is the practice of treating all blood and OPIM as if they are contaminated or infectious.
- Biosafety while working with humans is the consistent application
of safety measures to minimize or prevent exposure to the person, the
lab and building occupants, and the community andenvironment.
- All employees will use standard precautions while handling any type of human blood, human blood components, and materials made from human blood, or OPIM.
- These materials are to be handled as if known to be infected with HIV, HBV, or other bloodborne pathogens.
- All research involving the use of human blood, body fluids, and OPIM is carried out using Biological Safety Level BSL-2 practices, because it is unknown if these materials contain bloodborne diseases such as HIV, HBV, or HCV.
- For more information on Universal Precautions, visit the NIH/CDC website here.
Work Practices
Work practice controls are modifications of work procedures to reduce the likelihood of exposure to blood or OPIM. Use these work practice controls:
- Personal Protective Equipment (PPE).
- Hand Washing.
- Engineering Controls.
- Work Area Restrictions.
- Safe Specimen Handling and Transport.
- Disinfection, Decontamination, and Autoclaves.
- Biological Waste Disposal and Spill Response.
PPE
- PPE shall be provided at no cost to all employees at risk of occupational exposure to BBPs.
- Soiled PPE must not be taken home to launder. All garments that are penetrated by blood shall be removed and discarded immediately.
- PPE shall be chosen based on the anticipated exposure to blood or OPIM.
- PPE is appropriate only if it does not permit blood or OPIM to pass through or reach clothing, skin, eyes, mouth, or other mucous membranes under normal conditions of use.
- Wear appropriate gloves when there is reasonable anticipation of hand contact with blood or OPIM and when handling contaminated items.
- Never wash or decontaminate disposable gloves for reuse.
- Inspect gloves and replace if torn, contaminated, or if ability to function as a barrier is compromised.
- Gloves should be made of latex, nitrile, rubber, or other water impervious materials.
- Remove contaminated gloves carefully, avoiding touching the outside of the gloves with bare skin.
- Dispose of contaminated gloves in a proper container.
- Lotions and chemicals may break down gloves.


- Wear appropriate face and eye protection when splashes, sprays, or droplets of blood or OPIM pose a hazard to eyes, nose, or mouth.
- Safety glasses are minimum eye protection that should be worn when in laboratory and there is a potential for minor splash hazards with small amounts of liquid.
- Goggles offer eyes splash protection and should be used when handling corrosives, biological liquids, and large volumes of hazardous substance (i.e., more than one liter).
- Masks and face shields offer additional protection and must be used in combination with suitable eye protection (i.e., goggles and face shield).
- Refer to the LSUHSC PPE Policy for more information.
Eye Protection


Hand Washing
- All laboratories are required to have a sink available for hand washing.
- Wash hands for 15 seconds using warm water and mild (preferably liquid) soap.
- Rinse with warm running water.
- Dry with disposable paper towel.
- Alcohol-based hand sanitizers are an alternative to hand washing.
- Sanitizers are effective against common clinical microbes, but have not been tested against laboratory pathogens.
- Hand washing is preferred.

Engineering Controls
Engineering controls are physical controls that isolate or remove BBP hazards from the workplace. Examples include:
- Biological Safety Cabinets.
- Sharps Disposal Containers.
- Sharps with Engineered Sharps Injury Protection (SESIPs) and Needleless Systems.
- Splash Guards and Plastic Backed Absorbent Pads.
- Sealed Rotor Heads and Centrifuge Cups.
- Mechanical Pipetting Devices.


Biological Safety Cabinets
- Biological safety cabinets are a primary means of containment developed for working safely with infectious microorganisms.
- Provide containment of infectious aerosols, isolate the operator from the agent, and protect other personnel in the room.
- Must be certified annually, whenever moved, or after repair work has been performed.
- Contact EH&S for assistance with cabinet selection, certification and decontamination procedures, and proper placement in the lab.
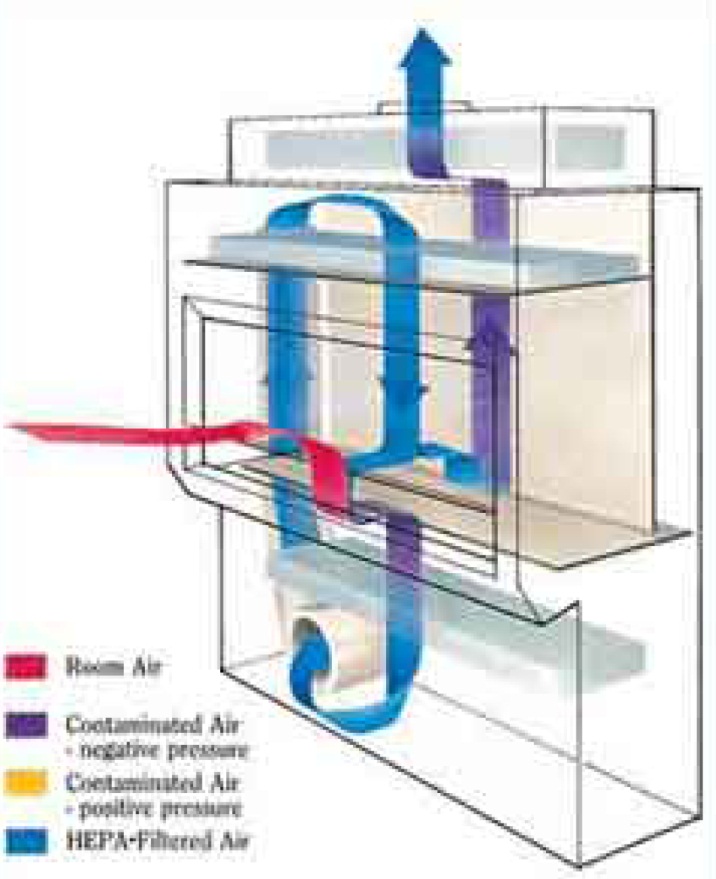
(Click or tap image for expanded view)
Sharps Containers
- Sharps containers must be used for disposing of all needles, scalpels and other sharps.
- Sharps must be placed in an appropriate sharps container immediately following usage and shall be placed as close to the procedure area as possible.
- Sharps containers must be non-breakable, puncture resistant, leak proof, sealable/closeable and labeled with the universal biohazard symbol.
- Sharps containers must be properly maintained and disposed of when ¾ full.

Sharps
- Sharps with Engineered Sharps Injury Protections (SESIPs) and needleless systems are recommended for work involving blood and OPIMs.
- Examples include syringes with protective shield, syringes with retractable needles, and blunt-tip blood drawing needles.
- Syringes which re-sheathe the needle, needleless systems, and other safety devices must be used whenever appropriate.
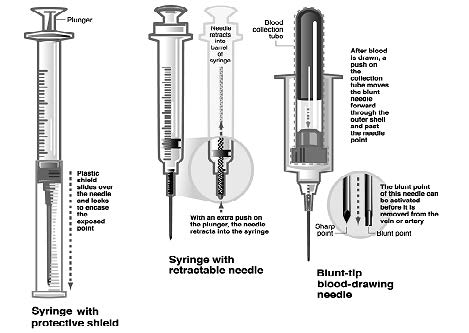
(Click or tap image for expanded view)
- Used disposable needles must not be bent, altered, broken, recapped, removed from disposable syringes, or otherwise modified.
- Always dispose of contaminated sharps in an approved, puncture-resistant sharps container.
- Dispose of container when it is ¾ full by sealing the container and placing in a biological waste box.
- An SOP for the safe use and disposal of sharps is available at the Biological Safety page of the EH&S website.
Splash Guards
- Splash guards and plastic backed absorbent pads must be used to contain the spread of blood and OPIM in the laboratory.
- Contaminated plastic backed absorbent pads shall be removed immediately or as soon as feasible after any spill of blood or OPIM as well as at the end of the workday.


Centrifuging and Pipetting
- Use sealed rotor heads and centrifuge cups to avoid accidental spills while performing routine centrifuge operations with material that potentially contains bloodborne pathogens.
- Mechanical pipetting devices must be used. Mouth pipetting is prohibited.



Work Area Restrictions
- Do not eat, drink, apply cosmetics or lip balm, or handle contact lenses in laboratories.
- Do not keep food and beverages in refrigerators, freezers, shelves, cabinets, or on counter tops or bench tops in laboratories.
- All procedures will be conducted in a manner that will minimize splashing, spraying, splattering, and generation of aerosol droplets of blood or OPIM.

Handling and Transport
- Blood or OPIM must be placed in a container that prevents leakage during the collection, handling, processing, storage, and transport of the specimen.
- The container used for this purpose will be labeled or color coded in accordance with 29 CFR 1910.1030 requirements and closed prior to handling.
- Any specimens that could puncture a primary container will be placed within a puncture-resistant secondary container.
- Personnel who pack and/or ship blood or OPIM off campus or
clinical site must complete LSUHSC EH&S On-line Training for
Shipping Biological Materials or have a current International Air
Transport Association (IATA) certificate.

Disinfection
- Disinfection is the process of reducing a contaminant load.
- Can be accomplished in the laboratory using a 70% solution of ethanol (EtOH) or a 10% solution of bleach (sodium hypochlorite).
- All works surfaces and materials should be disinfected before and after use.
- SOP for Laboratory Disinfection is available on the EH&S website
Decontamination
Decontamination is the process of removing biohazardous agents.
- Can be accomplished by physical or chemical means.
- Is typically done using an autoclave, utilizing high temperature and pressure.
- Aqueous solutions such as blood, urine, or microbial cultures must be autoclaved prior to disposal.
- An SOP for the safe use of autoclaves is available on the EH&S website.
- Place items in a secondary container made of stainless steel or autoclaveable plastic.
- Most pathogens and recombinant molecules are sensitive to temperatures above 121ºC for 20 or more minutes.
- Larger loads require more time and should be arranged in a way that allows for steam penetration (i.e., not too densely packed).
- Do not cap vessels or add excessive liquid to the load.
- Use caution when opening autoclave at the end of the cycle – steam is usually still in the chamber. Autoclaves

Biological Waste Disposal
- Use bioboxes with red biohazard liners to dispose of biohazardous waste.
- Use a sealable, leak-proof sharps container for all contaminated sharps (e.g., needle, scalpel, glass).
- Don’t overfill bioboxes (<¾) or make too heavy for transport.
- Seal sharps containers when ¾ full. Place sealed sharps container in biobox.
- Label bioboxes with supervisor’s name, building, room number, and contact phone number prior to placing in hallway for pickup.
- Bioboxes are available on the 2nd Floor MEB, 1st Floor CSRB, and Loading Dock area at the School of Dentistry. Sharps containers are available from Medical Stores.
- NEVER place chemicals, chemical bottles, radioactive materials, or other trash in biowaste containers.


Spill Response
Should a incident or spill occur involving blood or OPIM, do not attempt to clean it up without appropriate PPE. For major spills, contact University Police immediately, and notify Environmental Health and Safety to assist in cleaning up the spill.
- Secure the area, keeping all personnel clear of spill.
- Place paper towels, soaked in disinfectant, directly on spilled materials to prevent spread of contamination. To ensure adequate contact, surround the spill with appropriate disinfectant.
- Wash your hands thoroughly with soap and water immediately after the clean-up is complete.
- Refer to the Biological Spill Response Policy for additional spill response guidance.